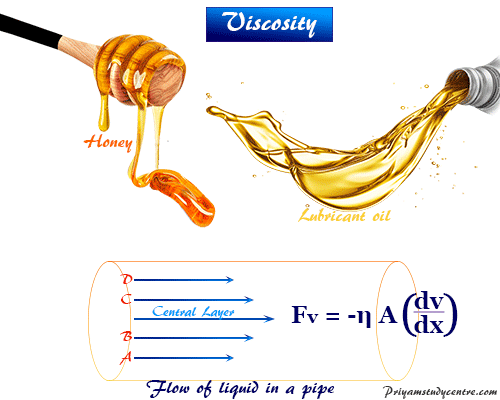
Rearranging the terms, gives the formula for dynamic viscosity:Ī Newtonian fluid is a fluid that obeys Newton’s law of friction, where viscosity is independent of the strain rate. Newton’s law of friction is an important equation relating to viscosity. This is due to hydrogen bonding between neighboring water molecules. The viscosity of water is low, yet it is higher than that of most other liquids made of comparable-sized molecules. On the other hand, water vapor viscosity increases as temperature increases. For example, water’s viscosity at 80 ☌ is 0.354 millipascals⋅second. Liquid water viscosity decreases as temperature increases. The dynamic viscosity of water is 1.0016 millipascals⋅second or 1.0 centipoise (cP) at 20 ☌. Its kinematic viscosity is 1.0023 cSt, 1.0023×10 -6 m 2/s, or 1.0789×10 -5 ft 2/s. And, of course, dynamic viscosity and kinematic viscosity have different units.

While the relationship is straightforward, it’s important to remember two fluids with the same dynamic viscosity values may have different densities and thus difference kinematic viscosity values. Absolute or dynamic viscosity is a measure of a fluid’s resistance to flow while kinematic viscosity is the ratio of dynamic viscosity to a fluid’s density. This is because intermolecular attraction doesn’t play a significant role in gas viscosity, but increasing temperature leads to more collisions between molecules. Increasing gas temperature increases viscosity. Gases also have viscosity, but the effect of temperature is just the opposite. In a liquid, increasing temperature decreases viscosity because heat gives molecules enough energy to overcome intermolecular attraction. For example, adding sugar to water makes it much more viscous.īut, temperature has the greatest effect on viscosity. The effect of adding molecules can be significant. The effect of pressure on liquids is small and often ignored. These include temperature, pressure, and the addition of other molecules. Viscosity looks at the difference in the rate of flow or deformation between between molecules a certain distance from a surface and those at the liquid-surface interface. They are only inhibited by their interactions with one another.
#Viscosity chemistry free
Meanwhile, molecules further from the surface are more free to flow. Basically, these molecules stick to the surface to a greater or lesser degree.
When you pour a liquid from a container, there is friction between the container wall and the molecules. As with friction between solids, higher viscosity means it takes more energy to make a fluid flow. Viscosity is friction between fluid molecules. An alternative and equivalent unit is pound-force-seconds per square foot (lbf In American and British engineering, another common unit is pound-seconds per square foot (lb This makes the viscosity of water at 20 ☌ about 1 cP or 1 mPa However, you’ll often see viscosity expressed in terms of pascal-second (Pa The SI unit for viscosity is newton-second per square meter (N Gas viscosity increases as temperature increases.Liquid viscosity decreases as temperature increases.Viscosity is a fluid’s resistance to flow.Common symbols for viscosity include the Greek letter mu (μ) and the Greek letter eta (η). Mistletoe berries yield a viscous glue, also called viscum. The word “viscosity” comes from the Latin word for mistletoe, viscum. A fluid with a high viscosity, such as honey, flows as a slower rate than a less viscous fluid, such as water. Viscosity is a fluid’s resistance to flow.īy definition, viscosity is a fluid’s resistance to flow or deformation. This entry was posted on Augby Anne Helmenstine (updated on November 18, 2021)
